
With such a large number of elements it is very difficult to study indivi dually the chemistry of all these elements and In a group, the electronegativity decreases as the atomic number increases, as a result of the increased distance between the valence electron and nucleus (greater atomic radius). Webchemical symbol, short notation derived from the scientific name of a chemical element e.g., S for sulfur and Si for silicon. Group 1A is also known as the alkali metals. What does a dream of crashing planets mean? These trends explain the periodicity observed in the elemental properties of atomic radius, ionization energy, electron affinity, and electronegativity. WebBy convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (18341907) in 1869, the table places elements into columnsgroupsand rowsperiodsthat share certain properties.These properties determine an elements Water is a compound. The elements are listed by atomic number (the number of protons in the nucleus), and elements with similar chemical properties are grouped together in columns. What are the properties of elements related to? Luster: Metals have the quality of reflecting light from their surface and can be One major classification of the elements is as metals, nonmetals, and metalloids. See more Encyclopedia articles on: Chemistry: General. Properties of an element are sometimes classed as either chemical or physical. 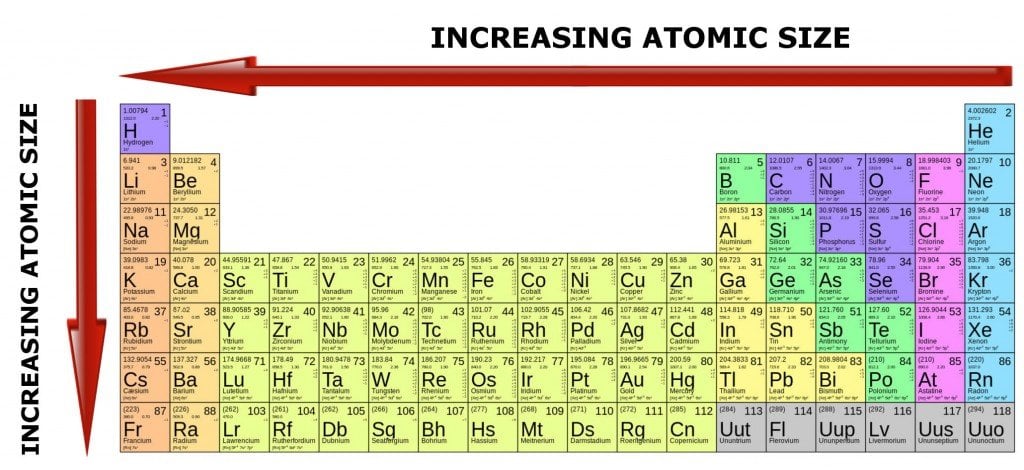
Generally, the atomic radius decreases across a period from left to right and increases down a given group. Which planet is yellow cloudy and super hot? WebFor example, many elements conduct heat and electricity well, whereas others are poor conductors. The exact value of sin 255 degree is a) (-sqrt3-1) / (2sqrt2) It is the energy change that occurs when an electron is added to a gaseous atom. Along these lines, an equivalent measured 3D shape of titanium, iron, and copper may all have a similar volume yet will have varying densities. Please refer to the appropriate style manual or other sources if you have any questions. chemical element, One of the 118 presently known kinds of substances that constitute all matter at and above the level of atoms (the smallest units of any element). 
Atomic radii decrease with the increase in the atomic number in a period. Electron Affinity Definition in Chemistry. These properties can be used to sort the elements into three classes: metals (elements that conduct well), nonmetals (elements that conduct poorly), and metalloids (elements that have properties of both metals and nonmetals). So many different types of gases and particles are present in the air, so air is not considered an element. The periodic table is also arranged by groups and periods. They write new content and verify and edit content received from contributors. Answer : The type of chemical bond form in molecule is covalent bond. an element that is shiny and that conducts heat and electricity well. Chemical bonds are the electrical forces of attraction that hold atoms or ions together to form molecules. WebYour most authoritative news analysis show, News File is live with Samson Lardy Anyenini. c)single replacement The Periodic Table can predict the properties of new elements, because it organizes the elements according to their atomic numbers. In each group (or column) of atoms, each atom will have the same number of valence electrons, and will thus have similar chemical properties. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.If you don't know how, you can find instructions. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Free Gift for you: Interactive Periodic Table In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. There are total 18 vertical columns on periodic table. Groups are the vertical columns on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
